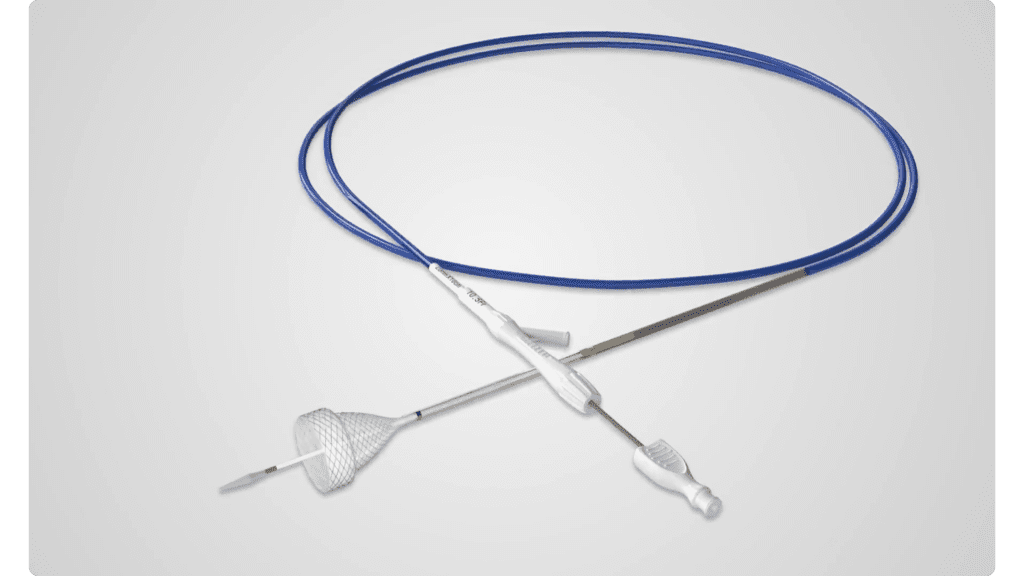
Versatility with the TaeWoong Esophageal TTS Stent
Pioneers of the TTS (through the scope technology). We are the first company to offer esophageal TTS stents in the US, bringing you the latest advancements in GI care, The TaeWoong Esophageal TTS Stent offers a variety of ways to treat patients.